Catalyst and substrate substituent effects were probed by means of systematic physical organic trend analysis. Little was previously understood regarding the structural origin of enantioselectivity in this system. As a validating case study, we selected an intramolecular dehydrogenative C-N coupling reaction, catalyzed by chiral phosphoric acid derivatives, in which catalyst-substrate association involves weak, noncovalent interactions. 
Here, we present a data-intensive method for deriving and then predictively applying a mechanistic model of an enantioselective organic reaction. 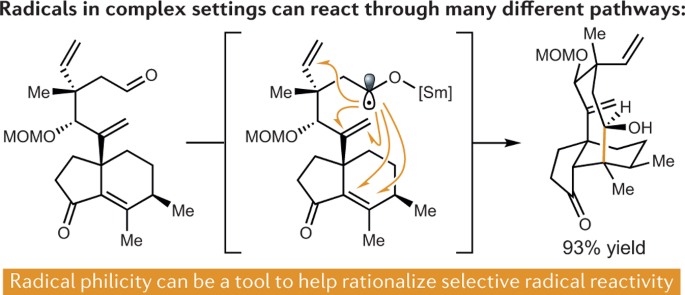
Knowledge of chemical reaction mechanisms can facilitate catalyst optimization, but extracting that knowledge from a complex system is often challenging.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
